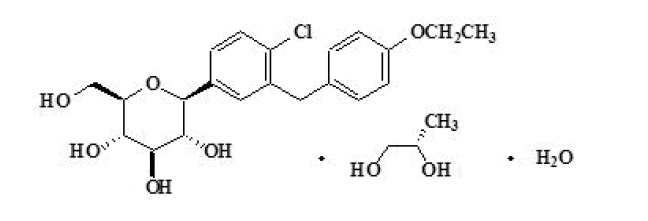
Dapagliflozin Propanediol Monohydrate
Therapeutic Category
- Anti-diabetic
Innovator Brand
GMP
DMF
Filed
Under Filing
Sample
Indication
Dapagliflozin as known from Brand Name Farxiga, is a sodium-glucose cotransporter 2 (sglt2) inhibitor indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
CTX Lifesciences, is one of the leading Active Pharmaceutical Ingredients ( API ) manufacturer and supplier globally for Dapagliflozin Propanediol Monohydrate with CAS# 461432-26-8. We are a leading exporter of Dapagliflozin Propanediol Monohydrate with CAS# 461432-26-8. We are a trusted supplier with API exports in more than 87 Countries including the US, Europe, Brazil, Latin America, China, Korea,Iran, Middle East and other emerging markets.
CTX Lifesciences is one of the leading API manufacturer & supplier of Dapagliflozin Propanediol Monohydrate with CAS# 461432-26-8. This product is provided from our GMP compliant plant with product made under complete cGMP conditions. We provided with documentation support Drug Master File(DMF). Our manufacturing plant at Surat has accreditation by US-FDA, EDQM , EMA , ANVISA, KFDA, MOH IRAN, Health Canada & Russian Authorities.
CTX has already filed Drug Master File with US-FDA for this product and will continue to offer O-Drug Master File(DMF) . This product is offered from our CGMP based facility based out of Surat, India.
CTX also offers Sodium/glucose cotransporter 2 inhibitors (also called gliflozins or flozins) , a class of medications that inhibit sodium-glucose transport proteins in the nephron (the functional units of the kidney). Please check them below.
CTX also offers other Dipeptidyl peptidase-4( DDP-4 ) inhibitor as mentioned below. Please check them out below.
It also offers an oxidative phosphorylation blocker i.e Imeglimin Hydrochloride
Disclaimer: CTX Lifesciences respects patent laws and conventions of pharmaceuticals as applicable in different countries.
API/Substances covered by patent are not offered to the countries where the patent law is in force. However, the final responsibility lies with the customer.